A new type of photovoltaic technology for converting solar energy into electrical energy is known as organic solar cells or OSCs. The technology makes use of a sandwich structure of metal electrodes/photoactive layers/transparent electrodes.
According to the sandwich structure described above, the active layer has not been properly defined. However, it is the core and most important part of the OSC and is one of the most important factors in determining the battery’s conversion efficiency.
The conversion efficiency is usually given by how n-type semiconductor acceptor materials are prepared and blended by the p-type conjugated polymer. Organic polymer materials are used as the active layer in organic polymer cells; these can be combined with flexible substrates.
These have many great characteristics, including flexibility, simple reparation process, lightweight and wide material sources.
Current important research topics include fullerene and its derivatives as electron acceptors and polymer/fullerene solar cells using conjugated polymers as electron donors.
The basic properties of fullerenes and polymers and their derivatives are optimized through molecular design strategies. These properties include the degree of crystallinity, molecular energy level and mobility of the two and absorption spectrum.
The energy conversion efficiency of the battery is also closely related to the crystallinity of the active layer material. DSC, or differential scanning calorimetry, can infer the material’s level of crystallinity and can directly measure the active layer material’s melting enthalpy.
This means that the user can infer the best ratio of electron donor and electron donor in the active material based on an estimation of the energy conversion efficiency.
This article uses PerkinElmer’s DSC 400 differential scanning calorimeter (shown in Figure 1) in accordance with the above test requirements to test the fullerene (PCBM and polymer (DBT).
The melting absorption peak, the crystallinity of the active layer materials and the regularity of the molecular arrangement are used to find the heating curve and melting enthalpy of the four active materials with different ratios.

Figure 1. PerkinElmer DSC 4000 differential scanning calorimeter. Image Credit: PerkinElmer, Inc.
Experimental Methods
The temperature rise and fall phases are 20ºC/min, the flow rate is 20 mL/min, the carrier gas is N2, the temperature scanning range is 30-300ºC and the initial temperature of the experiment is 30ºC.
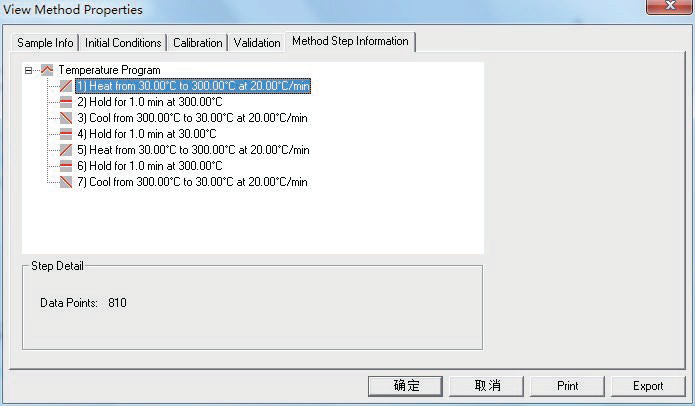
Figure 2. Experimental conditions for sample testing. Image Credit: PerkinElmer, Inc.
Figure 2 shows how the initial and final temperatures were placed in isothermal for 1 min in order to stabilize the sample temperature. The sample was warmed twice to eliminate the thermal history of the sample in order to get an accurate heat flow curve and enthalpy of melt of the sample.
The enthalpy of melt of the sample was calculated using the readings taken from the second warming curve. Four different mass fractions of fullerene (PCBM) and polymer (DBT) active later materials were configured prior to the experiment, with 10%, 20%, 40% and 50% mass fractions of polymer (DBT).
Experimental Results
Figure 3 shows the second warming curves of four active layer materials with different ratios of fullerenes (PCBM) and intercepted polymers (DBT). The melting enthalpy of the samples increased with the increase of DBT mass fraction, with the peak melting enthalpy of the four samples being measured at around 223ºC.
The molecular arrangement became more regular, and the internal compatibility of the material became better as the melting enthalpy of the active layer material of the battery continued to increase with the increase of DBT mass fraction.
However, the conversation efficiency of the battery may be affected by a high DBT mass fraction in the active material, which additionally affects the electron transfer efficiency.
This means the optimal ratio of fullerene (PDBM) to polymer (DBT) in the active materials should be deduced, taking into account the magnitude of the cell’s energy conversion efficiency.
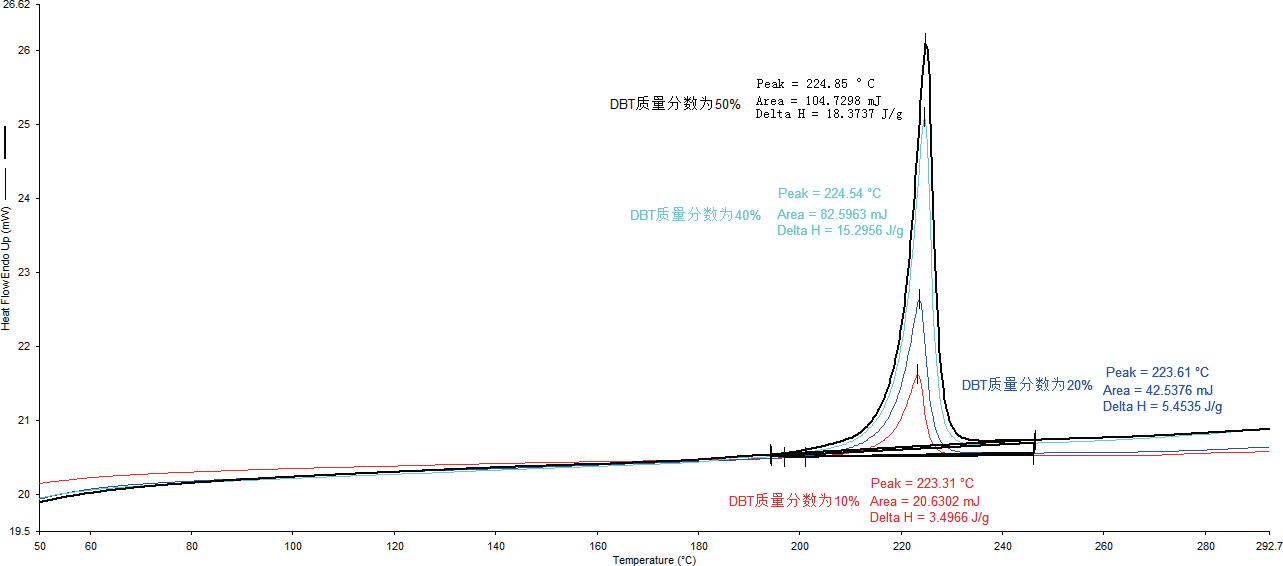
Figure 3. Melting curves and enthalpy of melting for the second warming of four different ratios of active materials. Image Credit: PerkinElmer, Inc.
Conclusion
DSC 4000 different scanning calorimeter was used in this article for the testing of the flow curve of the organic solar cell’s active layer materials. Additionally, the enthalpy of melting was calculated using the melting peak to determine the compatibility and molecular alignment of the material.
The data processing steps were simplified using the Peak Area calculation function in the software, improving the experimental efficiency.

This information has been sourced, reviewed and adapted from materials provided by PerkinElmer, Inc.
For more information on this source, please visit PerkinElmer, Inc.