Apr 18 2016
Specialized systems of materials, like those comprising of inorganic and organic thin films, are required to convert solar energy into solar fuels or electricity. Processes taking place at the junction of these thin films play a vital role in transforming the solar energy. A team from HZB, led by Prof. Emad Aziz, used ultra-short laser pulses and identified how boundary states are directly developed between the organic dye molecules and a zinc-oxide semiconductor layer, which temporarily traps the charge carriers. This explains the reason why zinc-oxide (ZnO) dye-sensitized solar cells have not been able to meet expectations. The results were delivered by the partnership between Monash University (Australia) and Freie Universität Berlin (FU Berlin) and Joint Lab partners Helmholtz Zentrum Berlin (HZB). These results have been featured online by Nature in the magazine Scientific Reports.
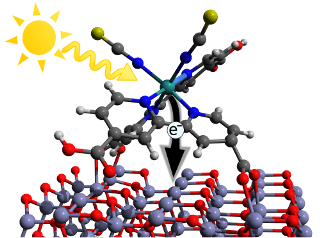 Illustration of the initial charge transfer step in a dye sensitized solar cell. A photon from the sun is absorbed and excites the dye molecule. Subsequently, an electron can is injected into the ZnO-Layer where it can be trapped by so called interface-states. (Credit: Mario Borgwardt/HZB)
Illustration of the initial charge transfer step in a dye sensitized solar cell. A photon from the sun is absorbed and excites the dye molecule. Subsequently, an electron can is injected into the ZnO-Layer where it can be trapped by so called interface-states. (Credit: Mario Borgwardt/HZB)
A wide range of materials help to transform the sun’s energy into solar hydrogen and electricity. Dyes used for a semiconductor material, like titanium dioxide (TiO2), are used by a specific class of organic solar cells. The dye molecules act as a translator for the solar energy. These molecules capture light and then inject electrons as carriers of free charges into the TiO2, resulting in the flow of current. However TiO2 is known to be far from ideal, and zinc oxide (ZnO) should be more suitable as an electrode material. This is due to the fact that the charge carriers are a lot more mobile in ZnO, and are able to flow more rapidly after the occurrence of charge separation. In addition, it is also possible to develop nanostructures capable of efficiently capturing sunlight in a simple manner by using ZnO.
Detailed investigation of excited states with ultrashort Laser pulses
Scientists have still not been able to produce ZnO solar cells better than those of TiO2. For the very first time, a team led by Emad Aziz has directly identified the reason for this and further examined it in great depth at the “Joint Ultrafast Dynamics Lab in Solutions and at Interfaces”. HZB along with FU Berlin operates the Joint Lab, which has a wide range of modern laser instruments including a time-resolved photoelectron spectrometer. This instrument is capable of generating ultra-short XUV pulses below 45 femtoseconds, which help to track the temporal and energetic development of excited states on ultra-short timescales.
Interface states as traps for charge carriers
Our measurements show directly for the first time that charge carriers are temporarily trapped by formation of an interface state between the dye and the semiconductor boundary layer. As a result, they are no longer immediately available as free charge carriers.
Mario Borgwardt, Doctoral Student, HZB
The electrons trapped within the interface remain stable for a long time. This feature increases the likelihood of them being lost again due to recombination. This results in decreasing the solar cell’s efficiency level.
A team from Monash University, Australia, headed by Prof. Leone Spiccia, supplied the samples used for the experiment. In 2015, Spiccia visited the university to receive the Helmholtz International Fellowship award from the Helmholtz Association. This collaboration indeed played a vital role towards bringing about the success of this project.
Helpful hints for the design of materials for energy conversion or storage
The work has led to a better understanding of the processes at the boundary layer between dye molecule and semiconductor. We have therefore been able to understand how dye and semiconductor materials communicate with one another. This enables us now to devise approaches for improving the communication in a direct way. That is important not only for the design of dye-sensitized solar cells, but also in order to be able to develop systems of materials for photocatalytic generation of hydrogen for storing solar energy as hydrogen fuel.
Emad Aziz, Professor, HZB