Dec 5 2017
Background
Industrial ethanol, a first-generation biofuel, is synthesized from foodstuffs such as maize, and hence presents considerable worry in relation to a probable future food shortage. Hence it would be wise to synthesize ethanol from non-food biomass such as waste paper, weeds, paper cup, and so on—that is, second-generation biofuel ethanol.
Solvents known to be used for synthesizing second-generation biofuel ethanol are extremely poisonous to microorganisms. To eliminate such extremely poisonous solvents, performing complex procedures such as separation by centrifugation, washing with water, compression, and so on is mandatory. Consequently, the energy retrieved from the ethanol synthesized by this process is less than the energy needed to synthesize it, which means a negative energy balance, that is, greater ethanol synthesis imposes more pressure on the environment. This challenge was deemed impossible to be overcome because a strong solvent was required to disintegrate recalcitrant plant materials such as cellulose, whereas such a strong solvent might kill microorganisms—including very tender and fragile cell membranes—that have significant roles in the fermentation needed for synthesizing ethanol.
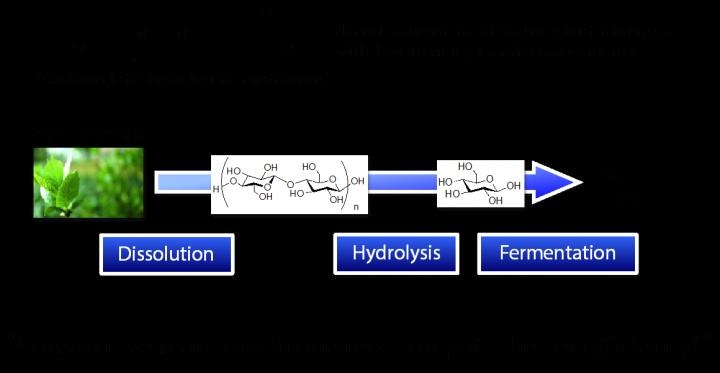 After dissolving plant biomass by the novel solvent, carboxylate-type liquid zwitterion, hydrolysis and fermentation were consecutively carried out in one reaction pot for conversion into ethanol. CREDIT: Kanazawa University.
After dissolving plant biomass by the novel solvent, carboxylate-type liquid zwitterion, hydrolysis and fermentation were consecutively carried out in one reaction pot for conversion into ethanol. CREDIT: Kanazawa University.
Results
In this research, scientists from Kanazawa University, Japan, worked to overcome this challenge. The team was successful in decreasing the toxicity to microorganisms by creating a new solvent, a carboxylate-type liquid zwitterion for dissolving biomass cellulose. The EC50, or a substance’s concentration needed to minimize the proliferation of Escherichia coli to 50%, was discovered to be 158 g/L for the recently created carboxylate-type liquid zwitterion. In contrast, the EC50 of ionic liquid, a traditional solvent of cellulose, was 9 g/L. This shows that the innovative carboxylate-type liquid zwitterion exhibits 17-fold lesser toxicity when compared to the ionic liquid.
By using E. coli with the ability to synthesize ethanol, fermentation potential was investigated and found to be nearly maximal in 0.5 mol/L carboxylate-type liquid zwitterion with a 21 g/L final ethanol concentration. In contrast, when the same experiment was conducted by using the ionic liquid, it synthesized just 1 g/L ethanol. Consequently, when fermentation was performed by using the carboxylate-type liquid zwitterion, 21 times more ethanol was synthesized than the amount synthesized with the ionic liquid.
In a different experiment, bagasse was adopted as a starting plant biomass for synthesizing ethanol by avoiding washing/separation procedures. By performing fermentation using the carboxylate-type liquid zwitterion, 1.4 g/L ethanol was synthesized. In contrast, no ethanol was synthesized when the ionic liquid was used, owing to its higher toxicity.
These experimental outcomes indicate that when the carboxylate-type liquid zwitterion was used, plant biomass could be transformed into ethanol inside a single reaction pot by avoiding the need for washing/separation processes. This seems to be a huge leap forward in the synthesis or use of second-generation biofuel ethanol by minimizing greater amounts of energy input.
Significance and future prospects
Apart from the first-generation and second-generation biofuel ethanol, a third-generation biofuel (a type of oil) can be synthesized by using specific algal species. To synthesize such a third-generation biofuel by using algae, polysaccharides such as cellulose (principal components of cell walls) must be dissolved. If dissolved polysaccharides can be transformed into ethanol, the energy efficiency will be considerably higher. Further advancement of this research will greatly enable the synthesis of second-generation as well as third-generation biofuel ethanol.
Moreover, the distinctive properties of the carboxylate-type liquid zwitterion have gained a considerable attention, and at present, three international partnerships are ongoing, one of which is with Rutgers University, United States.