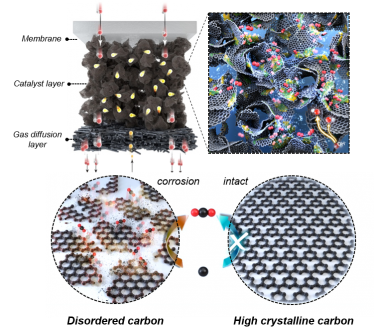
Image of nickel-iron-cobalt layered double hydroxide supported on hydrophobic crystalline carbon and image of crystalline carbon. Credit: Korea Institute of Science and Technology
Led by Dr. Yoo Sung Jong from the Hydrogen and Fuel Cell Research Center at the Korea Institute of Science and Technology (KIST), a research team has achieved a notable reduction in the production cost of green hydrogen.
They accomplished this by incorporating carbon support into an anion exchange membrane water electrolysis device, enhancing its performance and durability. Carbon supports are favored as they possess excellent electrical conductivity and a significant surface area, making them ideal for various electrocatalysts.
However, their usage has historically been limited because they tend to oxidize into carbon dioxide, particularly under high voltage and water-rich electrolysis conditions.
The research team synthesized a nickel-iron-cobalt layered double hydroxide material, which is a considerably more cost-effective alternative to iridium. They applied this material onto a hydrophobic carbon support and employed it as an electrocatalyst for the oxygen evolution reaction in anion exchange membrane electrolysis.
This catalyst demonstrated exceptional durability, thanks to its layered structure that interfaces with a hydrophobic carbon support and the nickel-iron-cobalt layered double hydroxide catalyst. Regarding carbon corrosion, the study revealed that the production of carbon dioxide during the corrosion process was reduced by over half.
This reduction was primarily attributed to decreased interaction with water, a critical factor in carbon corrosion. The amount of carbon dioxide generated during the corrosion process was found to be less than half due to the reduced interaction with water, which is a key factor causing carbon corrosion.
In the course of performance evaluation, the newly developed supported catalyst yielded remarkable results. It achieved a current density of 10.29 A/cm-2 in the 2 V range, surpassing the 9.38 A/cm-2 current density achieved by commercial iridium oxide.
Moreover, it exhibited impressive long-term durability, sustaining its performance for approximately 550 hours. A groundbreaking revelation emerged as well – a correlation between the electrolysis performance and the hydrophobicity of the carbon support was identified. This marks the first instance where it was demonstrated that the support's hydrophobic nature can significantly impact the performance of a water electrolysis device.
The results of this research confirm the applicability of water electrolysis devices on carbon supports, which have previously been limited in use due to corrosion problems, and it is expected that water electrolysis technology can grow to the next level if the research focused on catalyst development is expanded to various supports. We will strive to develop various eco-friendly energy technologies, including green hydrogen production.
Dr. Yoo Sung Jong Yoo, Korea Institute of Science and Technology
Journal Reference
Kim, M.-G., et al. (2023). Realizing the potential of hydrophobic crystalline carbon as a support for oxygen evolution electrocatalysts. Energy & Environmental Science. doi.org/10.1039/D3EE00987D.