Image Credit: Frontiers of Chemical Science and Engineering (2023).
Currently, overall water splitting (OWS) stands out as a viable method for hydrogen production. OWS, powered by renewable energy, enables hydrogen production through the hydrogen evolution reaction (HER) on the cathode.
The Faradic efficiency of hydrogen generation faces obstacles due to the anodic oxygen evolution reaction (OER), which is marked by high thermodynamic potential and sluggish kinetics. This challenge hinders the overall efficiency of the process.
As a result, there is a critical demand for the advancement of sophisticated electrocatalysts designed for the oxygen evolution reaction (OER) or other oxidation reactions, characterized by low thermodynamic potentials and rapid kinetics.
An emerging alternative gaining prominence is overall hydrazine splitting (OHzS), leveraging the anodic hydrazine oxidation reaction (HzOR). HzOR involves fewer electrons and exhibits faster kinetics compared to OER, making it a promising approach.
However, a significant challenge persists in synthesizing bifunctional electrocatalysts capable of facilitating both the hydrogen evolution reaction (HER) and HzOR with low overpotentials.
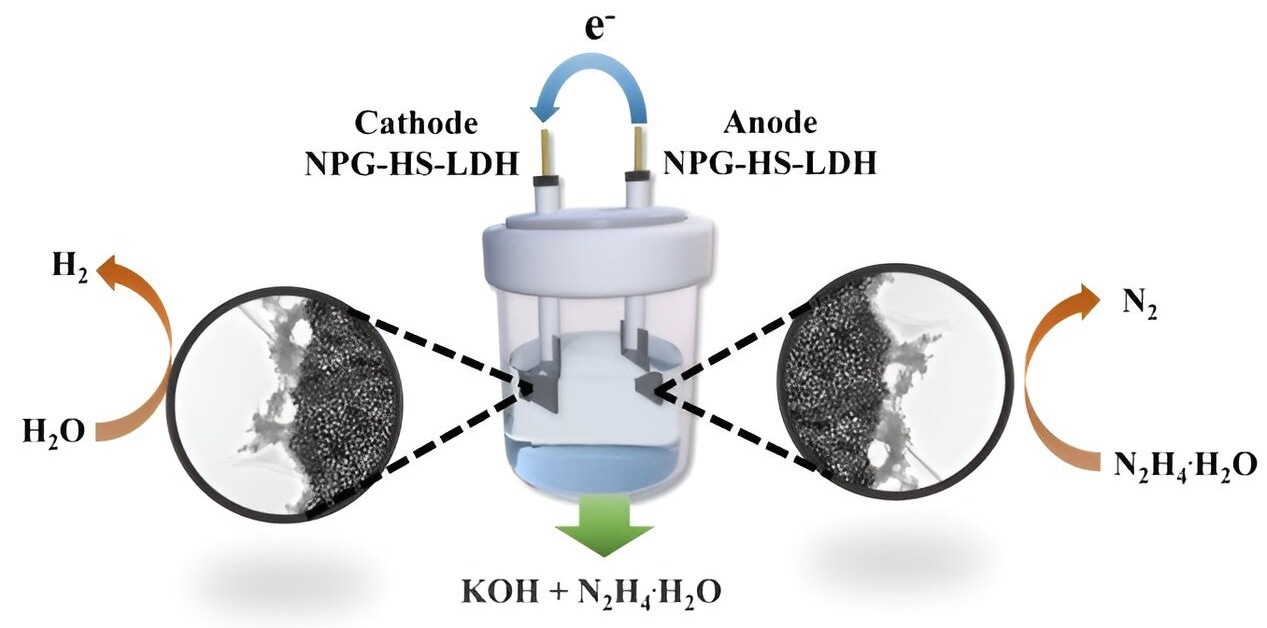
A recent breakthrough in the field has emerged from a research team in China. They introduced an innovative solution in the form of a two-dimensional multifunctional layered double hydroxide derived from a metal-organic framework sheet precursor. This material is bolstered by nanoporous gold, offering substantial porosity. The study was published in the Frontiers of Chemical Science and Engineering journal.
Remarkably, this electrocatalyst exhibits dual appealing activities for the hydrogen evolution reaction (HER) and hydrazine oxidation reaction (HzOR). In practical terms, the overall hydrazine splitting (OHzS) cell demonstrates superior performance, necessitating only a cell voltage of 0.984 V to achieve a current density of 10 mA∙cm-2.
This represents a significant improvement compared to the overall water splitting (OWS) system, which requires a higher voltage of 1.849 V.
Additionally, the electrolysis cell showcases remarkable stability by operating continuously for over 130 hours. This novel approach not only improves the efficacy of hydrogen production but also holds promise for fostering a more cleaner energy and sustainable future.
Journal Reference:
Qin, Y., et al. (2023) Multi-functional layered double hydroxides supported by nanoporous gold toward overall hydrazine splitting. Frontiers of Chemical Science and Engineering. doi.org/10.1007/s11705-023-2373-1.