Nov 21 2018
Researchers from Rutgers University–New Brunswick have created catalysts with the ability to convert carbon dioxide, the principal cause of global warming, into fabrics, plastics, resins, and other products.
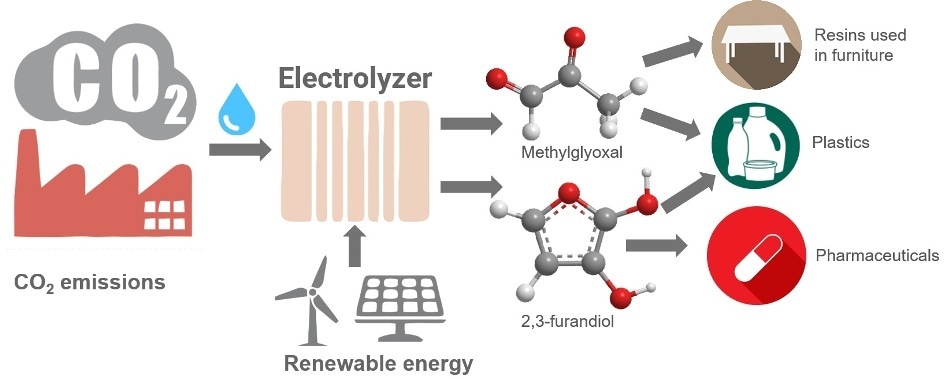 This image shows how carbon dioxide can be electrochemically converted into valuable polymer and drug precursors. (Image credit: Karin Calvinho/Rutgers University–New Brunswick)
This image shows how carbon dioxide can be electrochemically converted into valuable polymer and drug precursors. (Image credit: Karin Calvinho/Rutgers University–New Brunswick)
Apart from enzymes, the electrocatalysts are the first materials with the potential to convert carbon dioxide and water into carbon building blocks that contain one, two, three, or four carbon atoms with over 99% efficiency. It is feasible to use two of the products developed by the team—methylglyoxal (C3) and 2,3-furandiol (C4)—as precursors for adhesives, plastics, and pharmaceuticals. Methylglyoxal, which is safer, can be used in the place of toxic formaldehyde.
The discovery, which is based on the chemistry of artificial photosynthesis, has been discussed in the Energy & Environmental Science journal.
“Our breakthrough could lead to the conversion of carbon dioxide into valuable products and raw materials in the chemical and pharmaceutical industries,” stated Charles Dismukes, study senior author, who is a Distinguished Professor in the Department of Chemistry and Chemical Biology and Department of Biochemistry and Microbiology at Rutgers University–New Brunswick. He is also a principal investigator at Rutgers’ Waksman Institute of Microbiology.
Earlier, it was demonstrated by researchers that it is possible to electrochemically convert carbon dioxide into methane, methanol, ethylene, and ethanol with comparatively high yields. However, such production highly expensive to be commercially viable and inefficient, said Karin Calvinho, study lead author, who is a chemistry doctoral student in Rutgers’ School of Graduate Studies.
However, it is feasible to electrochemically convert water and carbon dioxide into a broad range of carbon-based products using five catalysts made of phosphorus and nickel, which are inexpensive and abundant, she stated. The choice of catalyst and other conditions govern the number of carbon atoms that can be stacked together to make molecules or even produce longer polymers. Typically, the length of the carbon chain is directly proportional to the value of the product.
Based on their study, the Rutgers researchers earned patents for the electrocatalysts and set up RenewCO2, a start-up company. The subsequent step is to gain further insights into the underlying chemical reaction, so that it can be employed to make other valuable products like diols, which are largely used in the polymer industry, or hydrocarbons that can be used as renewable fuels. The Rutgers scientists are designing, developing, and testing electrolyzers for commercial application.