Jan 16 2017
 (Credit: http://www.wiley.com/)
(Credit: http://www.wiley.com/)
Clean, fresh water coming directly from the tap is considered to be an absolute luxury. In developing countries, people often have no choice but to use a polluted river for drinking water. Water filters can help by rapidly converting polluted ground or surface water into safe drinking water.
Researchers have recently introduced a novel multifunctional composite material capable of removing organic, inorganic, microbial, and radioactive impurities from water. A report on this has been published in the journal Angewandte Chemie.
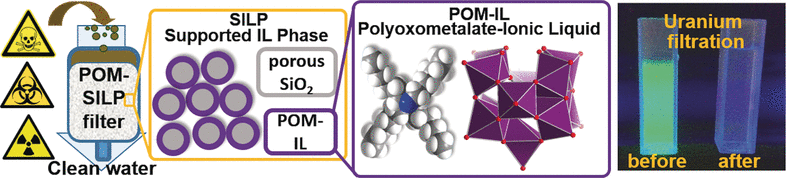
Generally, the water purification process involves a series of filters, where each filter is designed to remove one particular type of impurity. Conversely, the new filter material is an all-rounder. Scientists from the Universities of Ulm (Germany) and Zaragoza (Spain) have recently seized upon a comparatively new approach for designing materials capable of allowing molecular components to be assembled into multifunctional composites called supported ionic liquid phases (SILP) materials.
An ionic liquid is a salt melted at room temperature, making it a liquid without even being dissolved in a solvent. Adsorption of such an ionic liquid onto a solid substrate results in the formation of a solid composite material with properties that can be selectively altered through chemical modification.
The researchers, headed by Scott G. Mitchell and Carsten Streb, have now developed the first SILPs based on polyoxometallates (POM). POMs are known as molecular transition metal-oxygen clusters in which the metal atoms are bridged by oxygen atoms in order to produce a three-dimensional network.
Heavy metal ions are trapped by a binding site of these anions. The counterions selected by the researchers are voluminous tetraalkylammonium cations, which are known for their antimicrobial effect. The resulting ionic liquids are hydrophobic, immiscible with water, and develop stable thin layers on surfaces. The team used a porous silicon dioxide support to obtain dry, free-flowing powders that can be easily transported and handled.
The anions of the new composites consistently removed cobalt ions, chromium, copper, nickel, and lead in laboratory experiments. The silicon dioxide support directly trapped radioactive uranium in the form of UO22+. Similarly, the water-soluble blue trityl dye frequently used in the textile industry was also removed as a result of the lipophilic character of the ionic liquid. The growth of E. coli. bacteria is effectively halted by the antimicrobial cations.
The researchers anticipate that their new “POM-SILP” filter materials will form the foundation for the production of contaminant-specific chemically designed filter systems appropriate for the reliable purification of water in developing nations and remote areas. Additionally, the team hopes to use these new filter materials after chemical accidents and natural disasters.