A bioinspired nanosheet system mimics nature’s photosynthetic machinery to improve charge flow and unlock more stable, light-driven hydrogen production for future solar fuel technologies.
 Image credit: DimaBerlin/Shutterstock.com
Image credit: DimaBerlin/Shutterstock.com
A recent study in Advanced Energy and Sustainability Research presents a biomimetic strategy for artificial photosynthesis using ultrathin polydopamine (PDA) nanosheets. The system combines CdSe@CdS semiconductor nanorods with cobalt-based molecular catalysts in a redox-active polymer matrix. Inspired by the natural thylakoid membrane, PDA nanosheets enable efficient charge separation, suppress recombination, and support charge transport consistent with proton-coupled electron transfer. The system produces hydrogen under light and suggests a potentially scalable design toward future artificial leaf technologies, addressing ongoing challenges in solar-to-fuel conversion.
Biomimetic Design Tackles Charge Loss in Artificial Leaves
Artificial photosynthesis offers a promising way to produce sustainable hydrogen by converting solar energy into chemical fuels. However, current artificial leaf systems face several limitations. These include fast charge recombination in photosensitizers, degradation under illumination, poor coupling between electron and proton transport, and limited scalability of active interfaces. Together, these issues reduce efficiency and limit practical applications.
This work introduces a simplified artificial system based on polydopamine, a redox-active polymer with catechol and quinone groups. PDA has been shown to facilitate coupled electron-proton transfer processes in related systems, making it suitable for mimicking thylakoid membrane functions. The study builds a multifunctional platform by embedding CdSe@CdS nanorods as light absorbers and cobaloxime catalysts for hydrogen evolution within an ultrathin PDA nanosheet. This design integrates multiple essential functions into a single proof-of-concept, nanoscale system and aims to address key limitations in artificial photosynthesis at the materials level.
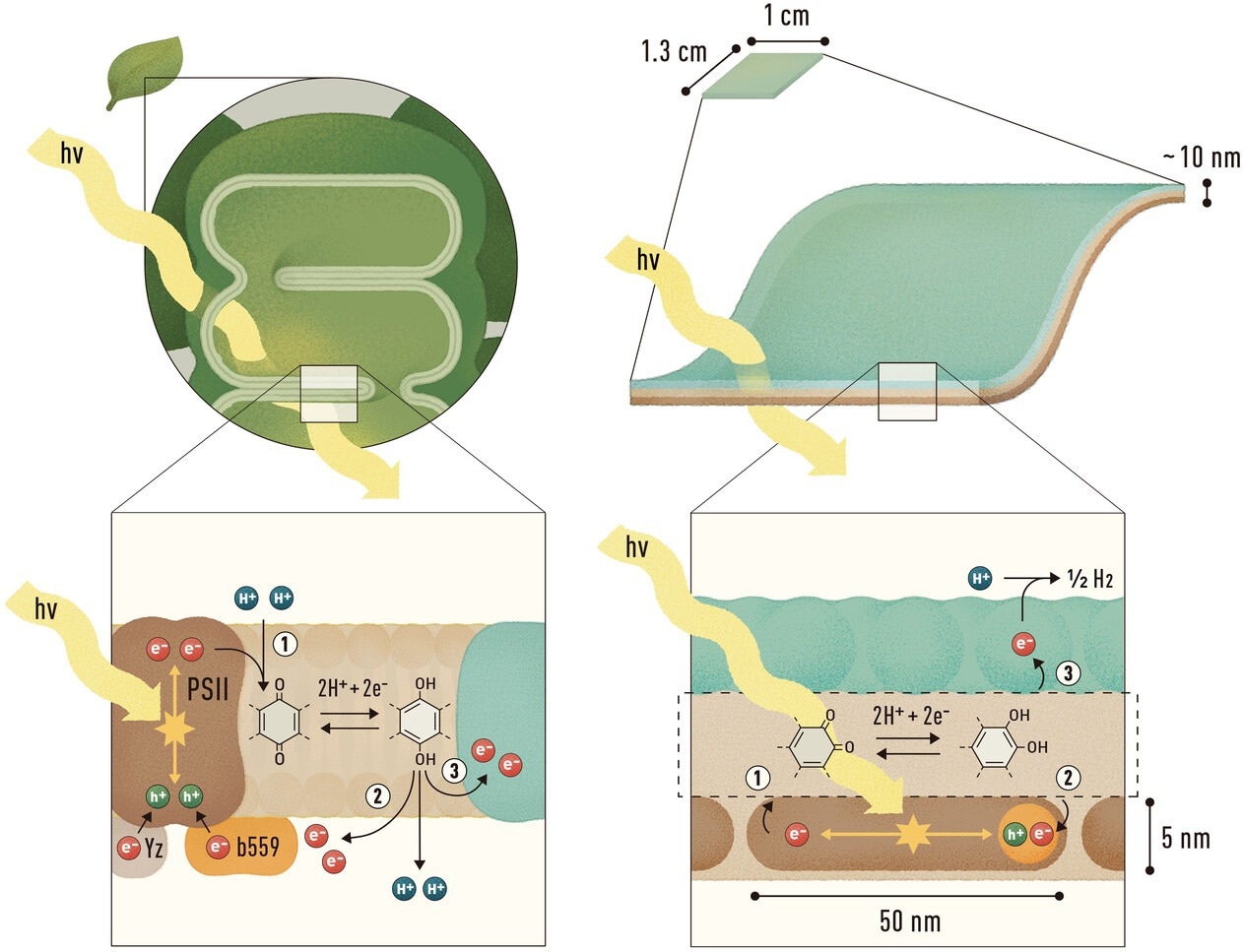 Comparison of natural and artificial photosynthetic systems: the left panel shows charge transfer processes in a thylakoid membrane, while the right illustrates a biomimetic polydopamine nanosheet embedding semiconductor nanorods and surface-bound catalysts, where light-driven electrons are transported through the polymer matrix to drive hydrogen evolution. Image Credit: Herberger, T., Kaplan, M., et al.
Comparison of natural and artificial photosynthetic systems: the left panel shows charge transfer processes in a thylakoid membrane, while the right illustrates a biomimetic polydopamine nanosheet embedding semiconductor nanorods and surface-bound catalysts, where light-driven electrons are transported through the polymer matrix to drive hydrogen evolution. Image Credit: Herberger, T., Kaplan, M., et al.
Spin-Coated Nanorods Form Light-Harvesting Foundation
The researchers designed a layered nanosheet system that integrates light harvesting, charge transport, and catalysis within a single platform. They deposited CdSe@CdS nanorods onto indium tin oxide substrates using spin coating. These nanorods act as efficient photosensitizers because of their favorable band alignment, strong light absorption, and ability to generate long-lived charge carriers.
Next, they formed ultrathin PDA films directly on the nanorods through electro-polymerization using cyclic voltammetry. This technique allows precise control over film thickness at the nanometer scale, which is essential for enabling efficient proton-coupled electron transfer. The resulting PDA matrix embeds the nanorods in a confined, redox-active environment that supports charge separation and transport.
Researchers modified the PDA surface with isonicotinic acid linkers, followed by the immobilization of cobaloxime hydrogen evolution catalysts to introduce catalytic functionality. These catalysts are known for their activity under mild conditions and compatibility with molecular systems. Surface characterization techniques such as X-ray photoelectron spectroscopy and time-of-flight secondary ion mass spectrometry confirmed successful catalyst attachment, chemical bonding, and uniform distribution across the nanosheet.
To evaluate performance, the researchers used a combination of photoelectrochemical measurements, transient absorption spectroscopy, and scanning electrochemical microscopy. These techniques provided detailed insights into charge-transfer dynamics, photostability, and hydrogen-evolution activity, enabling a comprehensive understanding of how the nanosheet functions under operating conditions.
Stratified Nanosheet Architecture Directs Electron Flow to Catalysts
Structural analysis revealed a well-organized architecture in which nanorods are embedded within the PDA layer, while catalysts are positioned at the outer surface. This spatial arrangement ensures that photogenerated electrons must travel through the PDA matrix to reach the catalytic sites, thereby promoting controlled, directional charge transport. Only the CdSe@CdS nanorods show unstable photocurrents due to rapid photooxidation. In contrast, PDA-integrated systems produce stronger and more stable photocurrents. The PDA matrix enables both anodic and cathodic responses, showing effective charge mediation and control over charge flow.
PDA facilitates the extraction of photoexcited electrons from the nanorods, as supported by spectroscopic and photoelectrochemical data, and helps reduce recombination by stabilizing charge-separated states. It also protects the nanorods from degradation, improving long-term stability. Film thickness plays an important role as thicker layers limit charge transport, while thinner layers enhance efficiency. Transient absorption spectroscopy confirms rapid electron transfer on picosecond timescales, indicating efficient charge extraction comparable in timescale to natural photosynthetic processes, although the fate of photogenerated holes remains unresolved.
Hydrogen evolution measurements confirm photocatalytic activity. The system produces hydrogen only under light and in the presence of catalysts. A delay in hydrogen generation suggests that charges may move through the PDA matrix via proton-coupled electron transfer rather than direct tunneling. Overall, PDA acts as both a charge mediator and a protective layer. It improves charge separation, increases stability, and enables sustained hydrogen production in a biomimetic system. The authors note that these measurements demonstrate functional performance under specific experimental conditions rather than providing direct benchmarking against other hydrogen evolution systems, and that they rely on a sacrificial electron donor rather than on overall water splitting.
Conclusion
This study establishes PDA nanosheets as a multifunctional platform for artificial photosynthesis. The system combines light absorption, charge transport, and catalysis within a single ultrathin structure, closely mimicking the natural thylakoid membrane. The redox-active PDA matrix is proposed to support proton-coupled electron transfer, reduce recombination, and protect photosensitizers.
The proposed nanosheet design is conceptually scalable, as its high surface-area-to-volume ratio allows stacking in principle, thereby potentially significantly increasing hydrogen production. This creates a potential path towards the development of compact and efficient solar fuel devices. This work shifts the design approach from complex multi-component systems to a unified material that performs multiple roles. This simplifies fabrication while improving performance and stability. Future research may explore different polymers, semiconductor materials, and scalable fabrication methods. It provides a strong foundation for next-generation artificial leaf technologies and highlights the value of combining materials science with biological inspiration.
Download your PDF copy by clicking here.
Journal Reference
Herberger, T., Kaplan, M., et al. (2026). Toward Artificial Thylakoids: Charge Transfer in Polydopamine Nanosheets for Photocatalytic Hydrogen Production. Advanced Energy and Sustainability Research, 7, e70180. DOI:10.1002/AESR.70180. https://advanced.onlinelibrary.wiley.com/doi/10.1002/aesr.70180
Disclaimer: The views expressed here are those of the author expressed in their private capacity and do not necessarily represent the views of AZoM.com Limited T/A AZoNetwork the owner and operator of this website. This disclaimer forms part of the Terms and conditions of use of this website.